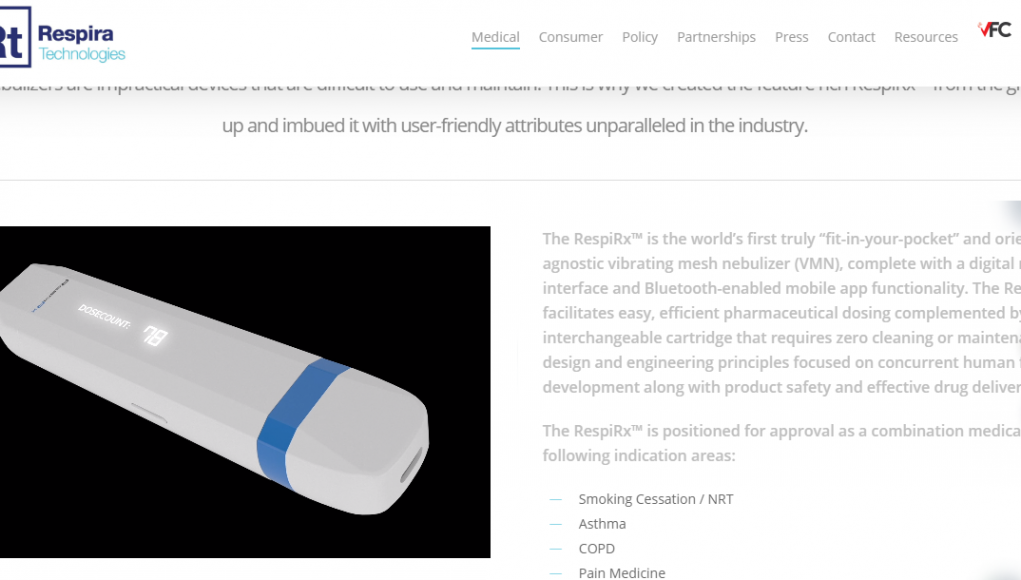
Respira Technologies Inc. is planning to submit a nicotine nebulizer to the Food and Drug Administration (FDA) for authorization as nicotine replacement therapy (NRT) by late 2021. Converting nicotine to aerosol, an article on the Ecomomic Times said that the device aims to disrupt the estimated $618 billion NRT market which is “dominated by decades-old gums and patches from pharma companies as well as tobacco companies’ more recent, ..controversial cigarette alternatives.”
Although the RespiRx device is similar to an e-cigarette in appearance, it works more like an asthma inhaler. Triggered by inhalation, the RespiRx should prove an easier transition from smoking than traditional NRTs such as gums and patches, as like e-cigarettes, it emulates the hand-to-mouth experience of using a cigarette.
The device should also be a favourite with tech-friendly individuals as it features blue-tooth wireless connectivity and its own app. Just like a Juul device that is in the works, the RespiRx aims to wean users off nicotine by providing the ability to meter and gradually reduce nicotine intake via the mobile phone app.
Voke
A similar device called Voke, was launched in the UK last April. This product had been in the making for years and has now been licensed as a medicine and a safer alternative to smoking by the Medicine and Healthcare products Regulatory Agency (MHRA).
Like RespiRx, Voke delivers rapid nicotine craving relief without heat, combustion or vapour and is renowned for being user friendly. However unlike the former, Voke looks a lot like a packet of cigarettes, including a rectangular box, and a plastic cigarette which slots into it. The price is also similar, with 20 charges costing £11.99. This is also likely to make this product a very effective NRT.
Most US Smokers Still Think That Snus is as Risky as Cigarettes